A recent study has revealed concerning levels of toxic PFAS "forever chemicals" in many popular...
Tag - FDA
Recalled Applesauce Linked to High Blood Lead Levels in 22 Children
The Centers for Disease Control and Prevention (CDC) is concerned about a potential link between...
WanaBana Apple Cinnamon Fruit Puree Pouches Recall
WanaBana USA is voluntarily recalling the WanaBana Apple Cinnamon Fruit Puree Pouches due to...
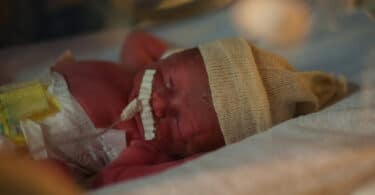
FDA Issues Warning About Probiotics for Preterm Infants
The U.S. Food and Drug Administration (FDA) is taking steps to protect public health by addressing...
FDA Approves Opill, the First OTC Birth Control Pill
Birth control can be instrumental in family planning. Unfortunately, obtaining it isn’t always...
FDA Announces Action Levels for Lead in Processed Baby Foods
The FDA has announced that it will take measures to ensure that lead levels in baby food are...
FDA Asks Parents To Throw Away Infant Head Shaping Pillows
Today the FDA issued a warning about infant head-shaping pillows. Infant head-shaping pillows...
What is Makena And Why Is It Possibly Being Taken Off The Market?
Introduced in 2011, Makena was approved under the accelerated approval pathway to 'reduce the risk...
FDA – Do Not Use Baby Neck Floats Due to the Risk of Death or Injury
The U.S. Food and Drug Administration (FDA) is warning parents, caregivers, and health care...
RECALL – Three Lots of Maple Island Inc. Parent’s Choice Rice Baby Cereal
Maple Island Inc. has issued a voluntary recall of three lots of its Parent’s Choice Rice Baby...
Johnson & Johnson recalls Aveeno and Neutrogena Spray Sunscreens After Benzene Detected
Johnson & Johnson Consumer Inc. (JJCI) is voluntarily recalling all lots of five NEUTROGENA® and...