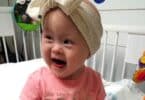
There is a seven-year-old boy lying in an intensive care unit in Memphis fighting as a virus takes control of his body leading to heart and kidney failure. Helpless family members watch the boy vomit blood several times a day. Each day they pray that one drug company will relent and provide the drug that may be the only way to save their son’s life.
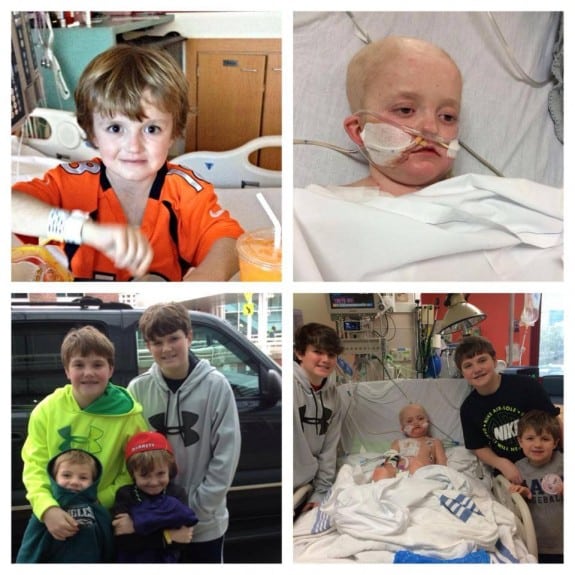
Image Via Facebook
The drug that could save the life of Josh Hardy is not yet in the market. The manufacturers Chimerix based in Durham, North Carolina are adamant that providing Josh with the drug could mean losing vital time, money and team effort in getting this drug onto the market.
The company feels helping Josh could mean hurting others.
Kenneth Moch, the president of the company, when asked what he feels about the seven year old battling for his life, replied promptly,
“Horrible,” he feels horrible and heartbroken.
But still they will not give the drug.
Josh’s family says,
‘We’re begging them. It’s called “compassionate use.”
According to Food and Drug Administration (FDA) if someone has a serious or a life threatening condition and have used all available treatment procedures but failed, they can request drug companies to provide drugs that may not be in the market yet, but may be the only way the person’s life can be saved. Such practice is called compassionate use of an experimental drug, still being tested in the lab and not yet approved by FDA.
In 2013 FDA approved 974 compassionate use of such drugs.
Manufacturing companies often say yes, but also say no and in the case of Josh, the drug is not being given.
“Our son will die without this drug,” Todd Hardy, Josh’s father told CNN. “We’re begging them to give it to us.”
The Hardy’s have now turned to social networking sites like Facebook and Twitter for help from the general public. Many concerned are asking Chimerix to relent and give Josh the life-saving drug.
Countless members of “Josh’s army” have responded with angry tweets to @chimerix, telling them to “open their hearts,” asking the executives how they can sleep at night. One angry tweet read,
“everyone is watching.”
Others have begun sending mails to board members. Chimerix executives say they have also received death threats.
Moch says he has read the tweets and is heartbroken, but the situation they are in, may not possibly be explained using twitter.
In simple terms he explains, “Chimerix is going full speed ahead to get the drug on the market hopefully by the end of 2016, and if they spend time and money on compassionate use cases, it would greatly hinder their effort to get the drug, brincidofovir, on the market and available to everyone.”
For compassionate use cases, the company would have to spend $50,000 per case as experimental drugs are not covered under insurance. More importantly is that the 50 person company would have to divert man power to handle the requests and then get the patient’s records and follow up with them, as required by the FDA.
“If this were just one patient wanting this drug, then this would be a very different question,” he said. “But it’s yes to all or no to all.”
But it is not that this particular drug was not given earlier for compassionate use.
Between 2009 and 2012 the company gave the drug to 451 patients but in those cases the use on patients was beneficialfor the Chimerix study. Currently the patient use is not helping the science go forward and therefore compassionate use has no purpose in speeding the process of getting the drug to the market.
Meanwhile, Josh struggles to stay alive every day. He was 9 months old when he was first diagnosed with a rare form of kidney cancer. Over the years, cancer turned up in his thymus, lung, and bone marrow, and each time Josh beat it.
But after a bone marrow transplant Josh’s immune system weakened and last year doctors detected an infection of adenovirus spreading in his body.
They gave him an antiviral drug, an intravenous form of brincidofovir, but it ravaged his kidneys.
Doctors at the St. Jude’s Children’s Research Hospital had been part of the study of this particular drug and assured the family that if the intravenous dosage had failed, the one given orally could be given without damaging the kidneys. They proceeded to ask the manufacturers Chimerix Inc. to use the experimental drug.
On February 12, the St. Jude’s doctors called a Chimerix executive, Dr. Marion Morrison, and asked for permission to use brincidofovir. She said no.
On March 5 they asked again. Two days later they got an answer by e-mail from another executive, Dr. Herve Mommeja-Marin, who said the company was not “in a position to provide drug for this and other subjects in similar circumstances due to a limited inventory and our limited resources.”
‘He holds our son’s life in his hands’ said the family.
On the other hand Moch says that he is a father himself and would have done the same thing to save his son’s life if he had been going through the same situation.
“There are no words to express our compassion for this young boy and his family and what they’re going through,” he said.
Art Caplan, a bioethicist at NYU Langone Medical Center told CNN there are many reasons why a company might say no to giving a drug for compassionate use.
“We can’t ask the company to turn into a philanthropy or their investors will back out,” he said.
He also adds that apart from the huge amount of $50,000 that investors might have to pay, compassionate case might make a drug look bad as the patient is already quite sick and the drug may make it worse. The manufacturing companies need to report back to FDA regarding the usage of the drugs and the poor outcome may go negatively in the future marketing of the drug.
Caplan suggests that Chimerix may still give the drug to Josh by giving the drug only in very dire cases and limiting the number of times they give it for compassionate use.
“They might want to open the door a little more broadly,” he said. “They might want to show a little compassion.”
But at present the company’s stand is a firm no.
“We’ve had employees who ask for the drug for family members who are close to death, and the answer has been no,” said Mommeja-Marin, the Chimerix executive.
The Hardys can’t believe the crossroads they are in.
“He holds our son’s life in his hands,” Todd Hardy said. “This is just beyond belief to me.”
At one end it is hard to believe that a company would go to such an extent to deny a child the only chance to live, on the other hand it also seems valid that the present no is a way to ensure that more like Josh are saved in future. But somehow, in the quest to mass market a life-saving drug, the manufacturer seem to have forgotten the basic essence of creating such drugs – to save lives no matter what. Josh is one life that needs saving now.